Electro-Exfoliation of Graphite to Graphene
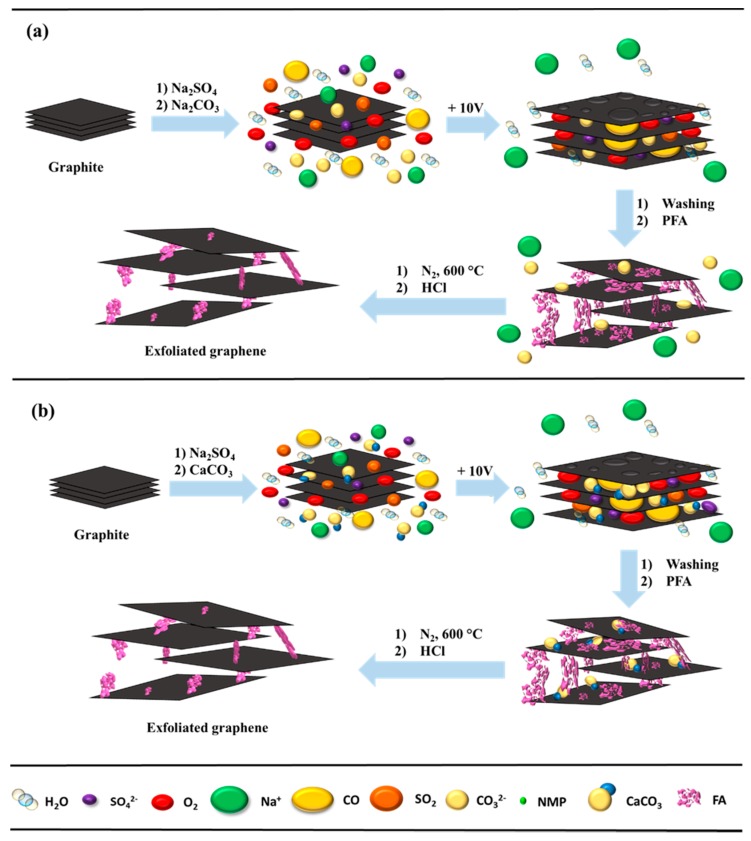
A group of scientists from the Faculty of Chemistry of the University of Mikołaj Kopernik in Toruń presented the results of their research of electro-exfoliation of graphite to graphene in an aqueous solution of inorganic salt. For the tests, graphite foil and graphite powder were used. Unlike other methods, e.g., chemical vapor deposition, epitaxial growth, and microchemical exfoliation, this approach is versatile in terms of chemical functionalization and a ording the possibility of high-volume production.
In conclusion, an accessible method was developed and demonstrated for obtaining few-layered graphene directly from graphite by electro-exfoliation and 3D stabilization by means of carbonized PFA as a stabilizer and certain inorganic carbonates as templates. The obtained samples can be treated as porous carbon sponges built of graphene flakes. The study indicates that the presence of carbonized PFA is critical for stabilizing the graphene and improving the surface parameters; the specific surface area increased several times compared to the starting material (graphite). The relatively high graphene purities and excellent stabilization with PFA show both methods to be promising ways of developing porous graphene. The morphological properties include the nanostructure of graphene powder with di erent sizes, characterized by SEM and HRTEM. The structural properties indicate a high quality for graphene, as determined by Raman spectroscopy. Low-cost and environmentally friendly production of such high-quality graphene is important, not only for future-generation electronics, but also for large-scale applications, such as composite materials, supercapacitors, fuel-cells, and batteries. Any practical application requires upscaling of the electrochemical arrangement and a cost calculation. The economic aspect of electrochemical exfoliation directly points to powdered graphite of high purity since its market prices are definitely much lower than the price of graphene foil. Experimental setup for exfoliation of graphite powder which was tested in the current project can be enlarged as single cell, but also small volume single cells may be used as a cell array. The presented electro-exfoliation approach has one general advantage over typical wet chemistry approaches: the inevitable chemical reagents i.e., electrolytes are common, inexpensive, and relatively easy to utilize. An upscale of the electrochemical cell will be the target of subsequent studies.
More:
https://www.ncbi.nlm.nih.gov/pubmed/31277243